NeuWave Collaboration: Shared Values Lead to Improved Patient Care – Department of Radiology – UW–Madison
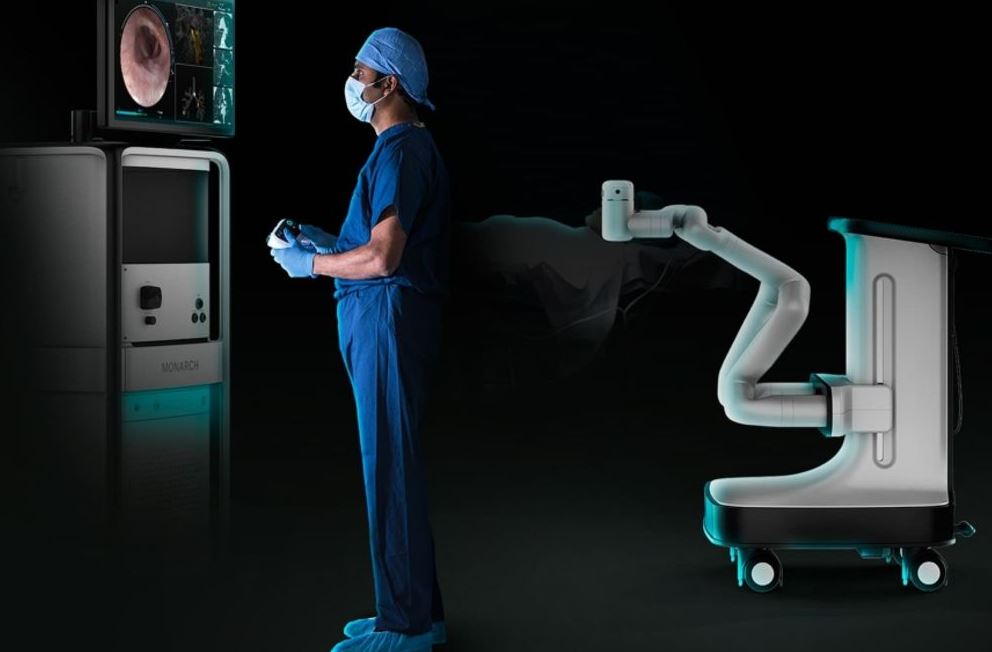
U.S. FDA Grants Ethicon Breakthrough Device Designation for Monarch-enabled NeuWave Microwave Ablation Technology | Legacy MedSearch - Medical Device Recruiters
October 24, 2018 NeuWave Medical, Inc. Dan Kosednar Director of Regulatory Affairs and Quality Assurance 3529 Anderson Street Ma
Global registry to collect real-world data on liver lesions ablated with the Neuwave microwave ablation system launches - 2829 Guardian Ln suite 150, Virginia Beach, VA 23452, USA